How Much Is a First Cycle Review ANDA Approval Worth to You?
At the recent GPhA meeting in Orlando, Florida, Dr. Kathleen Uhl from the FDA Office of Generic Drugs (OGD) spoke about the quality of Abbreviated New Drug Application (ANDA) submissions and highlighted the detriments of a poor quality submission. All would agree that a poor quality submission is costly for drug product development. Dr. Uhl discussed the various metrics for a first cycle quality ANDA submission, and presented the following:
- The number of Refuse to Receive (RTR) letters issued each fiscal year
- The first cycle review ANDA approval rate
- The number of ANDA review cycles prior to approval
In 2015 alone, approximately 24% of ANDA submissions resulted in Refuse to Receive letters. These letters were primarily associated with inadequate stability information and / or inadequate dissolution data. Many of these could have been avoided with closer attention given to the basic dissolution and stability requirements in the submission.
Existing ANDA Filing Guidance
The ANDA Filing Checklist is a great place to start when reviewing an ANDA prior to agency submission. The June 2013 Guidance for Industry ANDAs: Stability Testing of Drug Substances and Products and the September 2011 article published in the AAPS journal titled Dissolution Testing for Generic Drugs: An FDA perspective include the following basic recommendations:
Dissolution:
- Conduct comparative dissolution using a minimum of n=12 dosage units each of both test and reference products
- Generate dissolution data for products with samplings at multiple time points (3-4 time points at a minimum, equally spaced, excluding time zero) to characterize the dissolution profile
- Develop and communicate rationale for the Dissolution method used
- Use an FDA-recommended method. If not using an FDA-recommended method, then the agency expects to see both data from the sponsor’s method compared with the data using the FDA-recommended method
Stability:
- Submit data from a minimum of three batches which are manufactured and packaged using processes representative of the commercial processes
- Provide data from a minimum of 2 drug substance lots and additional data if more than one drug substance source
- Present a minimum of 6 months stability data that includes long-term and accelerated conditions at the time of submission
The Approval Facts
In her presentation, Dr. Uhl highlighted the number of review cycles required to gain ANDA approval, and how that number has changed over the years (Figure 1). What is the average number of CMC review cycles for an ANDA?
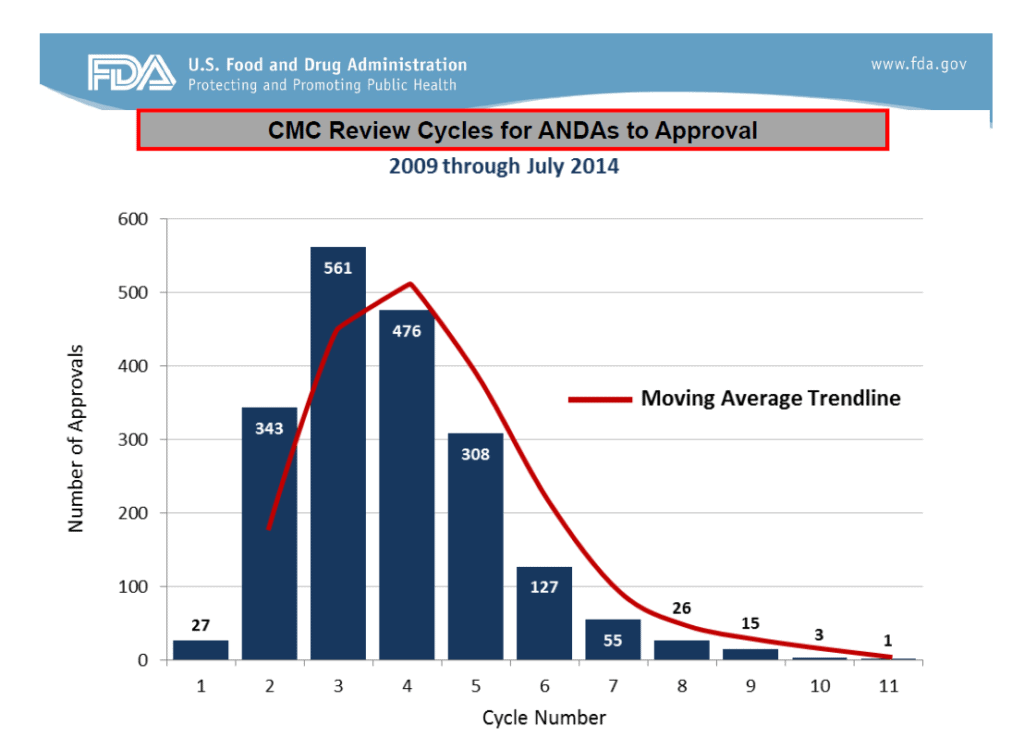
Since 2009, it reportedly takes an average of 4 CMC review cycles prior to an ANDA approval, as shown in Figure 1. Fixing the above CMC metrics is a sure way to reduce the number of review cycles.
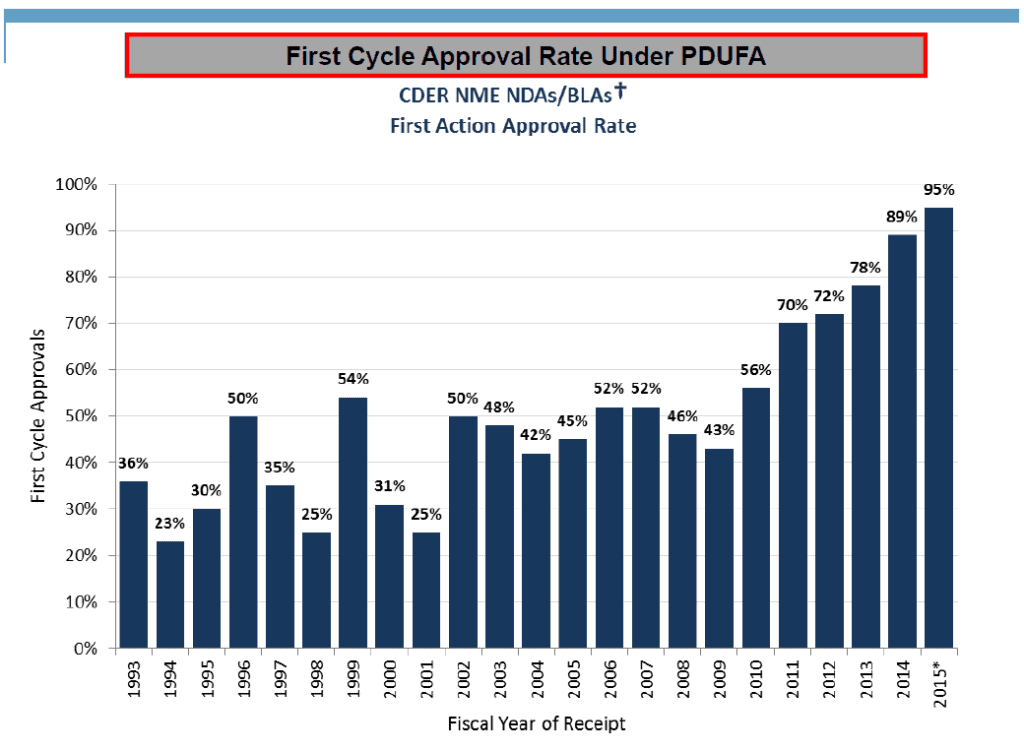
In contrast, an NDA is now approved on the first cycle review 95% of the time (Figure 2).
Additional Requirements
The basic requirements established in the above help guide a submission to filing, but don’t guarantee acceptance. This is where experience in the field helps, in understanding the nuances involved with the many more requirements based on the dosage form, route of administration, and active pharmaceutical ingredient.
Premier Consulting can provide expert guidance through the regulatory maze that leads to a first cycle ANDA approval, saving time and money, therefore improving a product’s return on investment. For more information about services Premier Consulting can provide in an ANDA and NDA application, visit our Services page or Contact Us for more information.