Improving Drug Development ROI in 2017
Time to Pick the Low-Hanging Fruit: Improving Drug Development ROI in 2017
With forecasts of decreasing peak sales for late pipeline drugs, a logical way to increase the return on investment (ROI) for pharmaceutical companies is to develop products with lower research and development (R&D) costs. How can this be achieved in an environment with increasing regulatory hurdles and clinical development costs?
R&D Returns for Large Pharma
Since 2010, Deloitte LLP and GlobalData have been tracking the R&D performance of 12 leading global life science companies by their R&D spend. In the most recent annual update, the authors included an additional cohort of 4 mid- to large-cap companies that have experienced recent rapid growth. Among the major findings of the report were:
- High-performing companies tend to have a consistent focus on fewer therapeutic areas resulting in greater R&D returns (more on this later).
- Mid- to large-cap* companies are delivering higher R&D returns than the larger companies. The difference was attributed to factors such as less operational complexity and the ability to make swifter data-driven decisions.
*The 4 “Mid- to large-cap” companies were “selected based on perceived recent performance and pharmaceutical R&D spend. These companies all fall within the top 25 pharmaceutical companies based on R&D spend(in)g for 2012 to 2014.”
- There may be a decrease in confidence in traditional R&D processes at larger companies. The authors based this on the finding of increased dividends returned to shareholders at the expense of investment in R&D, product licensing, and company acquisitions.
- Since 2010, the decline in forecast peak sales of assets (late stage pipeline products under development) had the greatest negative impact on R&D returns. The authors believe that it is unlikely that peak sales will reach the levels achieved in the past decade. Therefore, to get a higher ROI, they suggest that companies need to continue focus on:
- optimizing program and project costs
- developing an agenda to accelerate development timelines
- simplify the core processes and systems across R&D
- a consistent approach to therapeutic area investment.
Some good news to come from the report was that innovation levels were high. This was based on FDA reports of record numbers of approvals of new drugs, orphan designations, and breakthrough therapies for 2016. The report also cited data from the Pharmaceutical Research and Manufacturers of America that > 7000 drugs and treatments are in development globally. It is therefore reasonable to assume that new treatment options will continue to enter the market.
So how can a pharmaceutical company use these data to improve their ROI?
The 505(b)(2) Pathway Reduces Cost and Time to Market
Regular readers of this blog will be familiar with our core message that drugs approved via the 505(b)(2) pathway typically have reduced costs and accelerated development timelines compared with the 505(b)(1) pathway for new drugs. For those who have only tuned in more recently, the 505(b)(2) approval pathway is used to approve drugs for which some studies or data are already available thereby reducing the number of studies that a sponsor must conduct. So where are the numbers to back this up?
Let’s first consider that a significant portion of R&D costs cover products that fail in development. What are the chances that a 505(b)(1) vs. 505(b)(2) product will succeed?
A 2016 report by BIO (Biotechnology Innovation Organization), Biomedtracker, and Amplion tracked 9,985 clinical and regulatory transitions over the last 10 years from 7,455 development programs of 1,103 companies in their Biomedtracker database. When the authors compared the probability of approval success for new molecular entities (NMEs) with that of products that were neither NMEs nor generics [i.e., 505(b)(2)s], the results speak for themselves. As shown in the table below, at every stage of development, 505(b)(2) products have a greater chance of a successful approval than 505(b)(1) products.
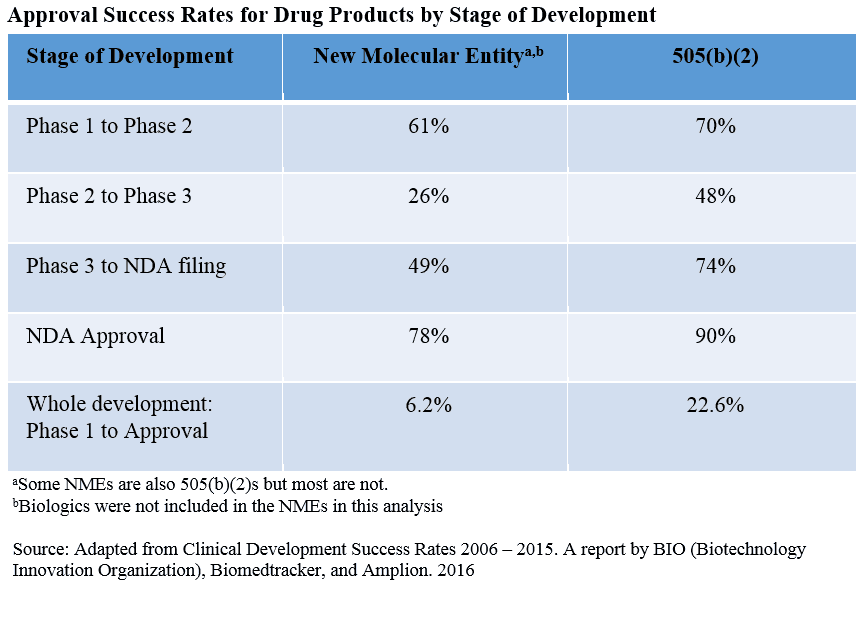
Even at the NDA stage, the probability of success is markedly greater for a 505(b)(2) product. Why is this? Products developed for approval via the 505(b)(2) pathway can be significantly de-risked with optimal regulatory interactions. This means that not only are less data/studies required for approval, but that if regulatory interactions are conducted properly, greater FDA feedback can be obtained earlier in the development process. In Premier Consulting’s experience, this greatly de-risks the development and approval processes.
The 505(b)(2) pathway has been growing in popularity as Sponsors learn of the benefits in reducing the size and scope of their development program. In 2015, FDA approved 73 NDAs for drugs (NMEs + non-NMEs but not biologicals), 44 of which were 505(b)(2) approvals (60%). While the number of NDAs has been steadily increasing over the last decade, so has the number of 505(b)(2) approvals.
Focus on Core Therapeutic Area/s
The Deloitte report noted that companies that have a consistent focus on fewer therapeutic areas enjoyed greater R&D returns than companies with more diverse portfolios. The authors believed that this was related to the deep knowledge and expertise that a company accumulates when it focuses on specific diseases or mechanisms of action over time. They postulated that constantly changing therapeutic area requires a higher investment to achieve similar returns. They also believe that companies with focused therapeutic areas negotiate more effectively with regulators.
Premier Consulting has noted in previous blogs that familiarity with FDAs therapeutic Divisions improves the quality of the regulatory action, and reduces unpleasant surprises at milestone meetings such as Pre-IND and Pre-NDA meetings.
Premier Consulting can help in several ways:
- Firstly, we can perform analyses on approved products in our proprietary 505(b)(2) database to help your company select therapeutic areas that best match your development and business goals.
- Secondly, we apply our familiarity with FDA Divisions to your product to map out a development program that closely aligns with FDA expectations for your target area. With as many as 6 FDA meetings per month, our cumulative knowledge is up-to-the minute with what each Division expects.
- Thirdly, our experience with therapeutic areas includes oversight/conduct of relevant clinical and nonclinical studies.
- Finally, our Product Ideation services result in constant analysis of commercial opportunities and evolving unmet medical needs in many therapeutic areas.
To learn more about how to improve your ROI or how to utilize our knowledge of your therapeutic area read more here or contact us.
Author:
Angela Drew, PhD
Product Ideation Consultant