2020 505(b)(2) NDA Approvals: A Year in Review, and What a Year It Was!
In 2020, CDER approved 68 NDAs that used the 505(b)(2) pathway, representing important advances in patient care across a wide range of therapeutic areas.
505(b)(2) Drug Improvement Approvals by Year
In spite of an unusual year due to the COVID-19 pandemic, NDA approvals via the 505(b)(2) pathway increased 6% from 64 in 2019 to 68* in 2020 (Figure 1).
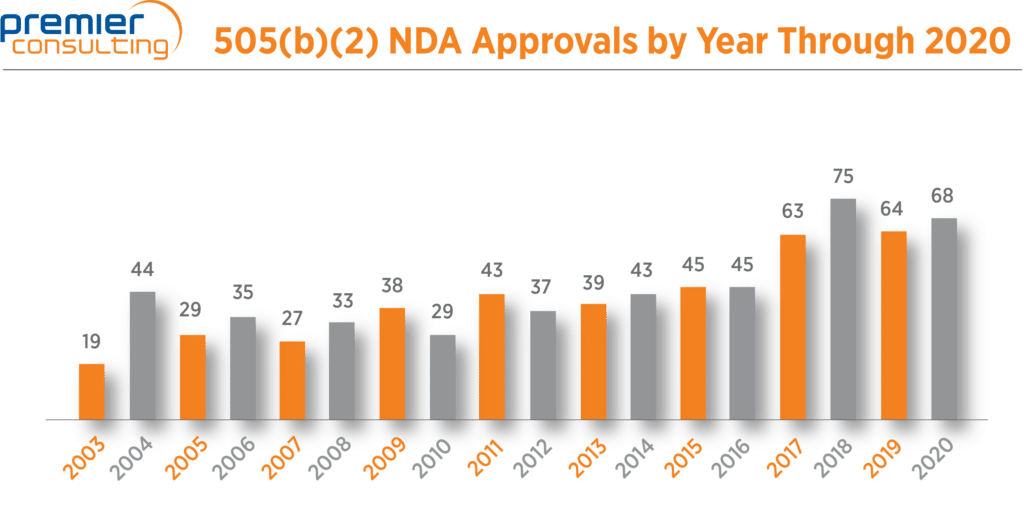 Figure 1. 505(b)(2) NDA Drug Approvals.
Figure 1. 505(b)(2) NDA Drug Approvals.
*This excludes BLA approvals and medical gas products approved through the FDA’s Center for Drug Evaluation and Research (CDER).
CDER Approvals — 505(b)(1) vs 505(b)(2)
Overall, NDA approvals granted by CDER held steady at 114 NDAs, the same as 2019. Drug improvement approvals via the 505(b)(2) regulatory pathway continue to account for well over half (60%) of all NDA approvals (Figure 2).
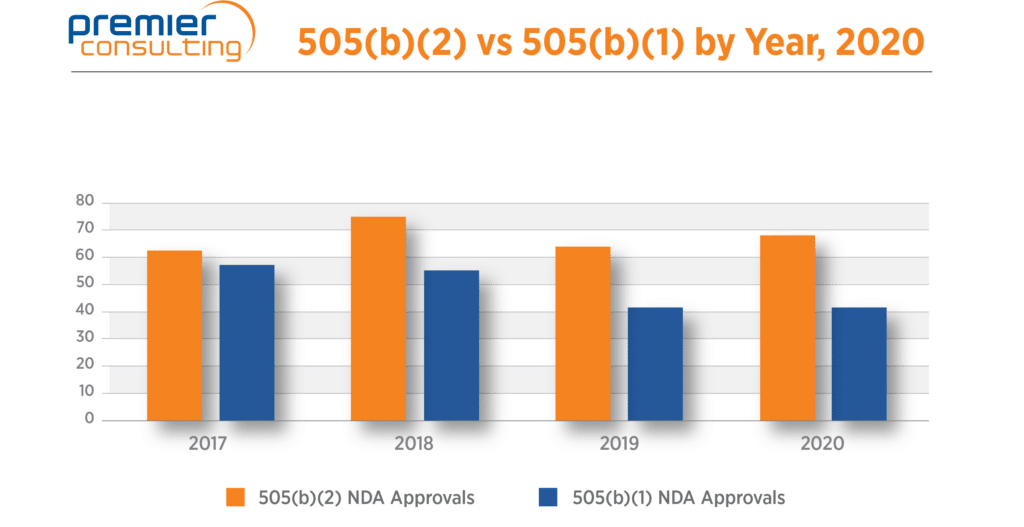 Figure 2. CDER Approvals for the 505(b)(1) and 505(b)(2) regulatory pathways.
Figure 2. CDER Approvals for the 505(b)(1) and 505(b)(2) regulatory pathways.
505(b)(2) NDA Review Times
The median review time for NDAs increased by one month from 10 months in 2019 to 11 months in 2020. More than half (55%) of all 505(b)(2) NDA approvals had review times 12 months or less, down from 71% in 2019. Only six 505(b)(2) NDAs had review times of six months or less, two of which were priority review products.
| 2017 | 2018 | 2019 | 2020 | |
| Median Review Time (Months) | 10 | 13 | 10 | 11 |
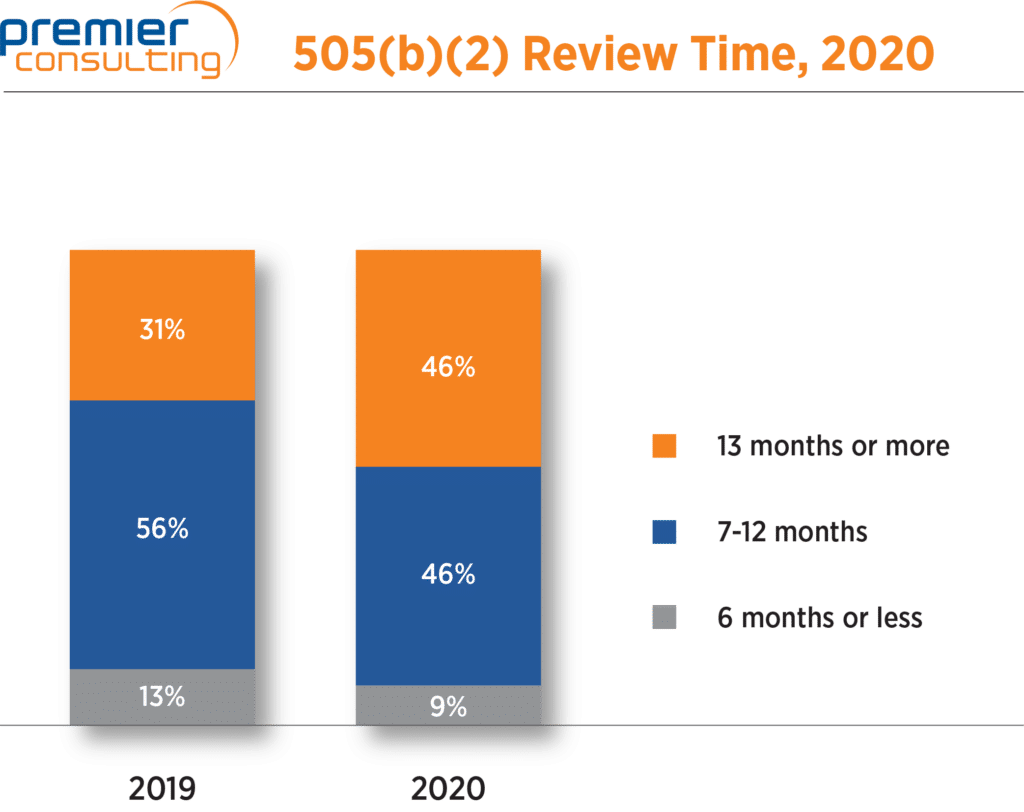 Figure 3. 505(b)(2) NDA Review Times.
Figure 3. 505(b)(2) NDA Review Times.
505(b)(2) Submission Classification Types: New Molecule Entity Submissions Continue to Grow
Though Type 5 (new formulation or other differences) and Type 3 (new dosage form) NDA submissions together continue to account for more than half of all 505(b)(2) approvals, Type 1 (new molecular entity) submissions increased sharply from 5% in 2019 to 18% in 2020 (Figure 4). As we’ve touched on previously on this blog, these approvals emphasize the potential for sponsors to achieve NME classification and associated marketing exclusivity through the 505(b)(2) pathway.
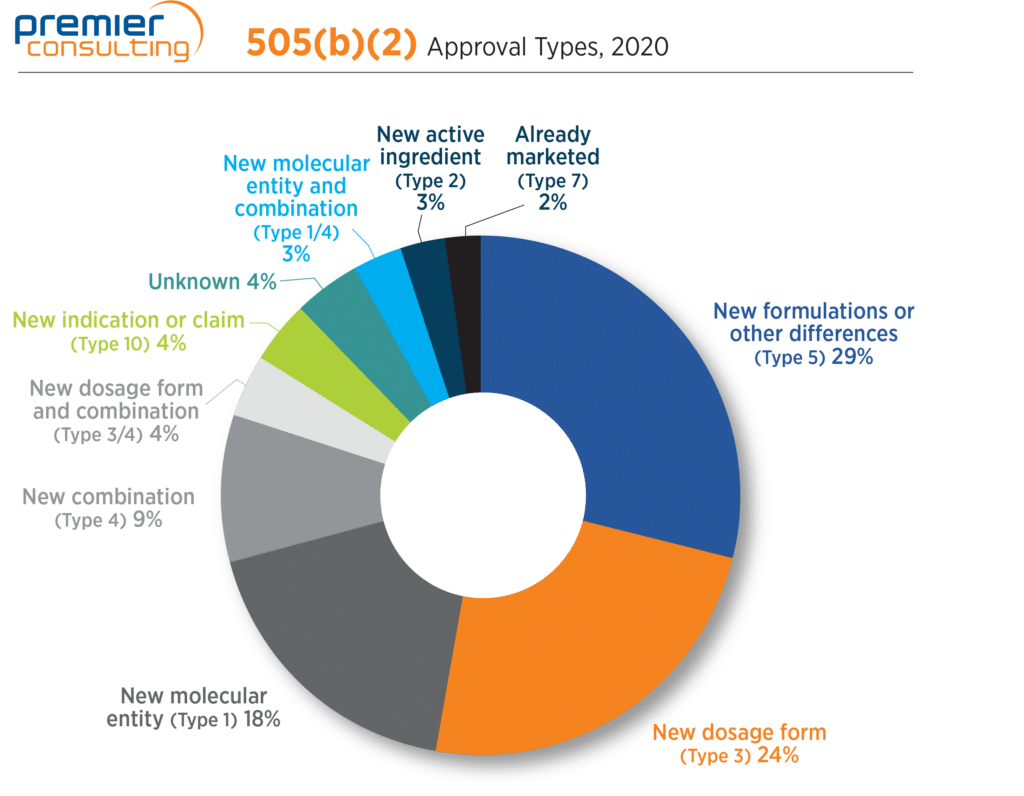 Figure 4. 2020 505(b)(2) Approvals by Type.
Figure 4. 2020 505(b)(2) Approvals by Type.
Rare Disease 505(b)(2) Approvals
Development strategies that leverage the 505(b)(2) regulatory pathway can be particularly relevant with rare disease patient populations, for which full Phase 1-3 clinical development programs may not be feasible. As shown in Figure 5, more than one third (36%) of NDA approvals for orphan drugs were developed via the 505(b)(2) pathway.
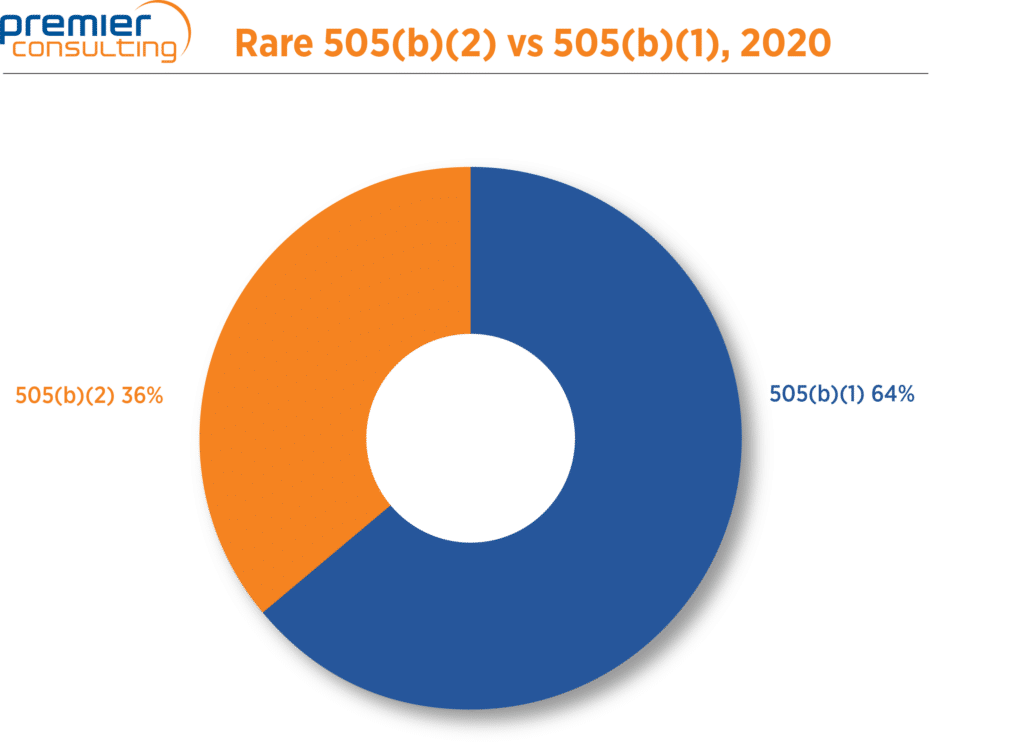 Figure 5. 2020 CDER NDA Approvals for Orphan Drugs.
Figure 5. 2020 CDER NDA Approvals for Orphan Drugs.
About the data
Data in this post comes from Premier Consulting’s proprietary 505(b)(2) database and from the FDA’s public databases including the Orange Book and drugs@FDA. Our 505(b)(2) database is maintained thanks to regulatory scientist Seth DePuy, PhD, in close collaboration with Angela Drew, PhD, Product Ideation Consultant.
Information is current as of April 7, 2021. Note that review documents are not yet available to confirm the regulatory approval pathway of some approved NDAs.[/vc_column_text][/vc_column][/vc_row]